 
Notifying the FDA of shortages of or problems with face masks, surgical masks, or respirators.Purchasing face masks, barrier face coverings, surgical masks, and respirators during the COVID-19 pandemic.Manufacturing and importing face masks, barrier face coverings, surgical masks, and respirators during the COVID-19 pandemic.Emergency Use Authorizations for face masks, surgical masks, and respirators.Using face masks, barrier face coverings, surgical masks, and respirators.The basics on face masks, barrier face coverings, surgical masks, and respirators.The FDA regularly updates its communications about face masks, surgical masks, and respirators, including the answers to frequently asked questions on this page. To help expand the availability of face masks, barrier face coverings, surgical masks, and respirators, the FDA is providing certain regulatory flexibility for the duration of the COVID-19 public health emergency, as described in the Enforcement Policy for Face Shields, Surgical Masks, and Respirators During the Coronavirus Disease (COVID-19) Public Health Emergency and Enforcement Policy for Face Masks and Barrier Face Coverings During the Coronavirus Disease (COVID-19) Public Health Emergency, and has issued emergency use authorizations (EUAs) for face masks, surgical masks, and respirators that meet certain criteria. The information provided may be useful to manufacturers and importers of face masks, barrier face coverings, surgical masks, and respirators, as well as health care facilities and health care personnel. Non health care use of face masks and respirators intended to limit industrial or general exposure to non-infectious particles, such as during construction or other industrial use.Powered respirators, such as powered air purifying respirators (PAPRs).This page provides information on face masks, barrier face coverings, surgical masks, and respirators (filtering facepiece respirators, such as N95 respirators) intended for a medical purpose to assist in preventing the spread of infectious materials during the COVID-19 pandemic. Webinar on Guidances on COVID-19 Transition Plans for Medical Devices - April 18, 2023.Transition Plan for Medical Devices Issued Emergency Use Authorizations (EUAs) Related to Coronavirus Disease 2019 (COVID-19).Transition Plan for Medical Devices That Fall Within Enforcement Policies Issued During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency.In particular, for manufacturers planning to seek marketing authorization for their devices, the FDA recommends beginning work on a marketing submission, including a transition implementation plan, as described in the guidances. The FDA encourages stakeholders to review the two final guidances, attend the webinar, and reach out to the FDA if they have questions or concerns. Taking other actions with respect to these devices.Developing a transition implementation plan,.The guidances outline the FDA's general recommendations to transition from certain policies adopted and operations implemented during the COVID-19 pandemic to normal operations, including the FDA's recommendations for: United Canada Masks offer more than a layer of protection – they offer a chance to support multiple Canadian businesses and to work with a team you can rely on.MaThe FDA has finalized two guidances: Transition Plan for Medical Devices That Fall Within Enforcement Policies Issued During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency and Transition Plan for Medical Devices Issued Emergency Use Authorizations (EUAs) Related to Coronavirus Disease 2019 (COVID-19). By responsibly sourcing materials and the hard work of our dedicated team, we can make that happen every day for thousands of Canadians Canada-wide. Our focus has always been on ensuring that every one of our customers is completely satisfied with their experience and purchase. We can oversee each aspect of the manufacturing process, ensuring proper care is being taken to produce a quality end product that our customers will love. Learn How We Manufacture Our Products?īy keeping our supply chain local, we can keep the quality of our level 3 face mask product and the shipping time low. Designed and manufactured by Canadians for Canadians. 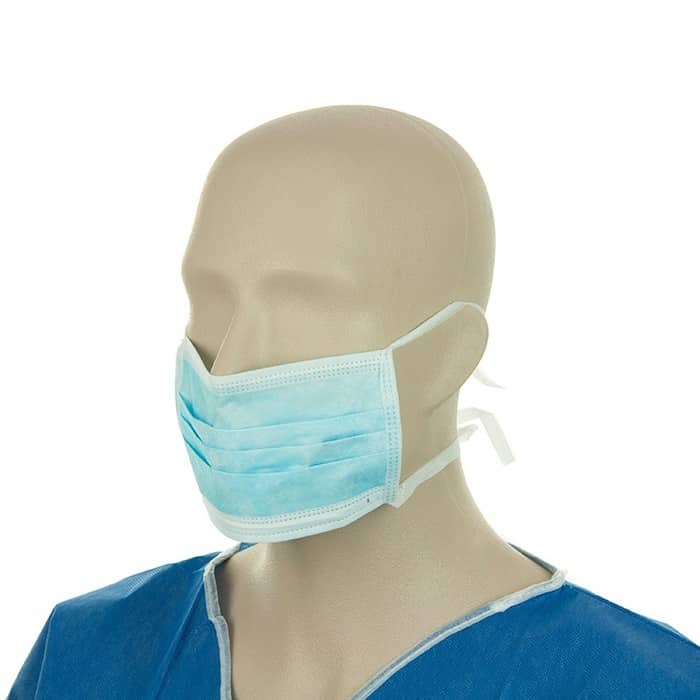
Our masks offer users a high breathability while exceeding ASTM F2100-20 standards. United Canada Level 3 Surgical Face Masks provide the highest protection without sacrificing comfort. Secure & Comfortable Fit level 3 face Mask Every order ships out of our Ontario, Canada, manufacturing facility. Medical Masks are manufactured in North America using entirely North American materials. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
